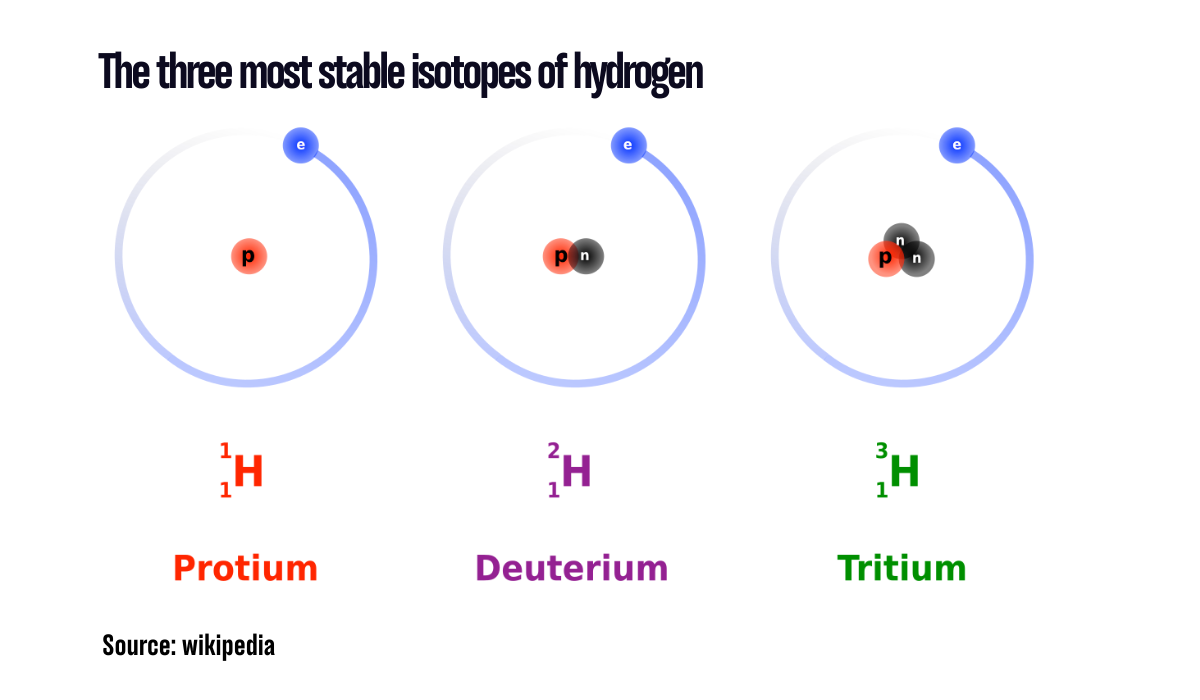
Nuclear ABC: I is for Isotopes
What People Get Wrong

Isotopes, especially radioactive ones, often carry an unfair reputation.
The misconception? All isotopes are dangerous, linked only to nuclear accidents, waste, or weapons.
This misunderstanding stems from a lack of awareness about the many beneficial uses of isotopes, the strict safety protocols in place, and the fact that not all isotopes are radioactive — and even those that are can be used safely.
Some believe that any exposure to a radioactive isotope is life-threatening, ignoring differences in radiation type, quantity, and context. Others think isotopes are just unwanted byproducts of nuclear waste, overlooking their intentional production for medicine, agriculture, and industry.
Nuclear Reality Check

In reality, isotopes — both stable and radioactive — are indispensable tools in medicine, science, and industry.
Medical isotopes like technetium-99m enable life-saving diagnostic imaging for millions each year, while iodine-131 treats thyroid disease effectively.
Agriculture uses isotopes to improve crop yields and control pests. Industry relies on them for material testing, leak detection, and quality control. Stable isotopes support environmental research, helping scientists study climate change, water cycles, and pollution.
When handled responsibly under regulation, the benefits of isotopes far outweigh their risks.
Facts That Matter

- 🏥 Medical Applications: From cancer diagnostics to treatment, isotopes save lives daily.
- 🌾 Agricultural Benefits: Enhance soil fertility, develop pest-resistant crops, and improve water use.
- 🏭 Industrial Uses: Detect structural flaws, prevent leaks, and ensure safety in infrastructure.
- 🌍 Environmental Research: Track climate trends, water movement, and pollution patterns.
- ⚡ Energy Production: Uranium-235 and plutonium-239 power low-carbon nuclear reactors.
- 🔬 Scientific Advancements: Support breakthrough research in physics, chemistry, and biology.